Integration and label transfer with Tabula Muris#
Following the general tutorial on atlas-level integration, here we show using scVI and scANVI for label transfer. In this setting we have:
Fully-labeled reference dataset
Unlabeled query dataset
We will demonstrate this functionality using Tabula Muris:
Tabula Muris Consortium. “Single-cell transcriptomics of 20 mouse organs creates a Tabula Muris.” Nature 562.7727 (2018): 367-372.
It is important to note that this tutorial covers de novo integration. With scvi-tools you can also perform online or sequential integration (i.e., reference mapping). Please see the scArches tutorial for more details.
Note
Running the following cell will install tutorial dependencies on Google Colab only. It will have no effect on environments other than Google Colab.
!pip install --quiet scvi-colab
from scvi_colab import install
install()
WARNING: Running pip as the 'root' user can result in broken permissions and conflicting behaviour with the system package manager. It is recommended to use a virtual environment instead: https://pip.pypa.io/warnings/venv
import os
import tempfile
import anndata
import matplotlib.pyplot as plt
import numpy as np
import pandas as pd
import scanpy as sc
import scvi
import seaborn as sns
import torch
from scvi.model.utils import mde
scvi.settings.seed = 0
print("Last run with scvi-tools version:", scvi.__version__)
Last run with scvi-tools version: 1.1.0
Note
You can modify save_dir below to change where the data files for this tutorial are saved.
sc.set_figure_params(figsize=(6, 6), frameon=False)
sns.set_theme()
torch.set_float32_matmul_precision("high")
save_dir = tempfile.TemporaryDirectory()
%config InlineBackend.print_figure_kwargs={"facecolor": "w"}
%config InlineBackend.figure_format="retina"
We supply Scanpy’s read function with a backup url, which covers automatic data download.
tm_droplet_path = os.path.join(save_dir.name, "TM_droplet.h5ad")
tm_facs_path = os.path.join(save_dir.name, "TM_facs.h5ad")
tm_droplet = sc.read(
tm_droplet_path,
backup_url="https://figshare.com/ndownloader/files/23938934",
)
tm_facs = sc.read(
tm_facs_path,
backup_url="https://figshare.com/ndownloader/files/23939711",
)
We subset to labelled bone marrow cells from female mice because it is a reasonably sized dataset that will allow us to validate our method’s ability to transfer labels from one dataset to another. In Tabula Muris, we are provided data collected using 10x Chromium, as well as SmartSeq2 data.
tm_droplet = tm_droplet[
(tm_droplet.obs.tissue == "Marrow")
& (~tm_droplet.obs.cell_ontology_class.isna())
& (tm_droplet.obs.sex == "female")
].copy()
tm_facs = tm_facs[
(tm_facs.obs.tissue == "Marrow")
& (~tm_facs.obs.cell_ontology_class.isna())
& (tm_facs.obs.sex == "female")
].copy()
tm_droplet.obs["tech"] = "10x"
tm_facs.obs["tech"] = "SS2"
tm_droplet.shape, tm_facs.shape
((11707, 20138), (4200, 22966))
Dataset preprocessing#
Normalize Smartseq2 matrix by gene length#
We apply gene-length normalization because the number of reads representing a transcript is proportional to the lenght of the transcript, unless UMIs are used (as in 10x). A discussion of this phenomenon can be found in this 2017 paper by Phipson B. Other than the gene length normalization, no other normalization is needed. scVI or scANVI is designed to handle sequencing depth and does not need cell-wise normalization. Normalizing and scaling the data will be detrimental to the performance of scVI and scANVI since they model explicitly the count data.
The gene length file here is computed by taking the average length of alal transcripts corresponding to a mouse gene recorded in the ensembl dataabase.
gene_len = pd.read_csv(
"https://raw.githubusercontent.com/chenlingantelope/HarmonizationSCANVI/master/data/gene_len.txt",
delimiter=" ",
header=None,
index_col=0,
)
gene_len.head()
| 1 | |
|---|---|
| 0 | |
| 0610007C21Rik | 94.571429 |
| 0610007L01Rik | 156.000000 |
| 0610007P08Rik | 202.272727 |
| 0610007P14Rik | 104.000000 |
| 0610007P22Rik | 158.750000 |
gene_len = gene_len.reindex(tm_facs.var.index).dropna()
tm_facs = tm_facs[:, gene_len.index]
assert (tm_facs.var.index == gene_len.index).sum() == tm_facs.shape[1]
tm_facs.X = tm_facs.X / gene_len[1].values * np.median(gene_len[1].values)
# round to integer
tm_facs.X = np.rint(tm_facs.X)
Dataset concatenation and gene selection#
Another important thing to keep in mind is highly-variable gene selection. While scVI and scANVI both accomodate for large gene sets in terms of runtime, we usually recommend filtering genes for best performance when the dataset has few number of cells. As a rule of thumb, performance starts to decrease when number of cells and number of genes are comparable. This point is emphasized in this comparative analysis of data integration algorithms for scRNA-seq data.
We perform this gene selection using the Scanpy pipeline while keeping the raw data in the adata.raw object. We obtain variable genes from each dataset and take their intersections via the Scanpy function.
adata = anndata.concat([tm_droplet, tm_facs])
adata.layers["counts"] = adata.X.copy()
sc.pp.normalize_total(adata, target_sum=1e4)
sc.pp.log1p(adata)
adata.raw = adata # keep full dimension safe
sc.pp.highly_variable_genes(
adata,
flavor="seurat_v3",
n_top_genes=2000,
layer="counts",
batch_key="tech",
subset=True,
)
Integration with scVI#
As a first step, we assume that the data is completely unlabelled and we wish to find common axes of variation between the two datasets. There are many methods available in scanpy for this purpose (BBKNN, Scanorama, etc.). In this notebook we present scVI. To run scVI, we simply need to:
Register the AnnData object with the correct key to identify the sample.
Create an SCVI model object.
scvi.model.SCVI.setup_anndata(adata, layer="counts", batch_key="tech")
scvi_model = scvi.model.SCVI(adata, n_layers=2, n_latent=30)
Now we train scVI. This should take a couple of minutes on a Colab session
scvi_model.train()
Epoch 400/400: 100%|██████████| 400/400 [03:51<00:00, 1.73it/s, v_num=1, train_loss_step=881, train_loss_epoch=1.05e+3]
Once the training is done, we can evaluate the latent representation of each cell in the dataset and add it to the AnnData object
SCVI_LATENT_KEY = "X_scVI"
adata.obsm[SCVI_LATENT_KEY] = scvi_model.get_latent_representation()
Finally, we visualize the learned scVI embeddings.
SCVI_MDE_KEY = "X_scVI_mde"
adata.obsm[SCVI_MDE_KEY] = mde(adata.obsm[SCVI_LATENT_KEY])
INFO Using cuda:0 for `pymde.preserve_neighbors`.
sc.pl.embedding(
adata,
basis=SCVI_MDE_KEY,
color=["tech"],
frameon=False,
ncols=1,
)
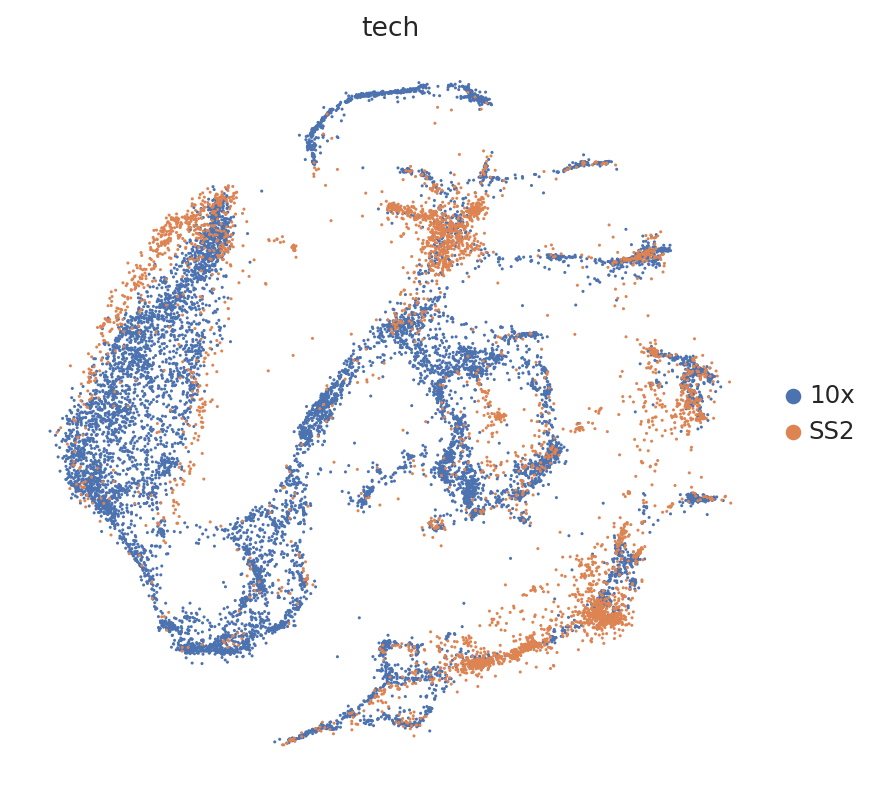
Because this combination of datasets is used for benchmarking purposes, we have access here to curated annotations. We can use those to assess whether the harmonization worked reasonably well.
sc.pl.embedding(adata, color="cell_ontology_class", frameon=False, basis=SCVI_MDE_KEY)
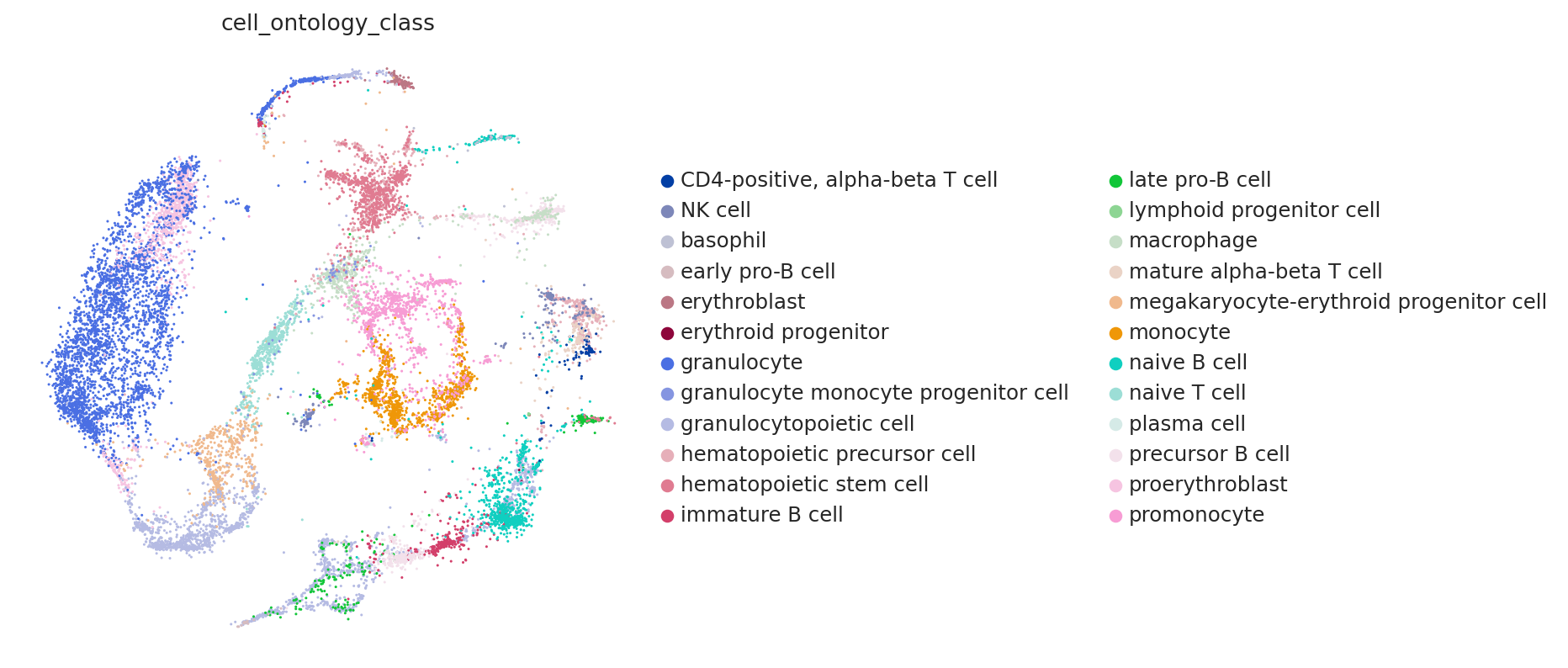
From a quick glance, it looks like the integration worked well. Indeed, the two datasets are relatively mixed in latent space and the cell types cluster together. A more refined analysis may be done at the level of markers.
Transfer of annotations with scANVI#
With scVI alone, we can train a classifier (e.g., RandomForestClassifer) on the latent representation of the labeled data and then obtain predictions for the query data. For example, we can imagine the 10x data is unannotated and then proceed to transfer labels using the latent reprsentation of the SS2 data.
We can also try to obtain a better latent representation/predictions by using the labels to inform the latent space. This is where scANVI comes in. scANVI uses semi-supervised learning to improve the model learned with scVI, allowing us to transfer our cell type knowledge from the reference to the query data. For this, we simply need to indicate to scANVI:
the sample identifier for each cell (as in scVI), which in this case is the technology (10x vs SS2)
the cell type, or an unnassigned label for each cell
scANVI labels#
We assume that the SS2 data is annotated and the 10X data is not. Only the labels of the cells from the labelled SS2 dataset will be kept in the adata.obs column 'celltype_scanvi'. All the 10X cells will have 'celltype_scanvi' of value ‘Unknown’.
SCANVI_CELLTYPE_KEY = "celltype_scanvi"
adata.obs[SCANVI_CELLTYPE_KEY] = "Unknown"
ss2_mask = adata.obs["tech"] == "SS2"
adata.obs[SCANVI_CELLTYPE_KEY][ss2_mask] = adata.obs.cell_ontology_class[
ss2_mask
].values
np.unique(adata.obs[SCANVI_CELLTYPE_KEY], return_counts=True)
(array(['CD4-positive, alpha-beta T cell', 'NK cell', 'Unknown',
'basophil', 'early pro-B cell', 'granulocyte',
'granulocyte monocyte progenitor cell', 'granulocytopoietic cell',
'hematopoietic stem cell', 'immature B cell', 'late pro-B cell',
'lymphoid progenitor cell', 'macrophage',
'mature alpha-beta T cell',
'megakaryocyte-erythroid progenitor cell', 'naive B cell',
'precursor B cell', 'promonocyte'], dtype=object),
array([ 107, 139, 11707, 26, 24, 773, 52, 67, 894,
281, 132, 13, 136, 150, 22, 740, 321, 323]))
Since we’ve already trained an scVI model on our data, we will use it as a pretrained model to scANVI.
scanvi_model = scvi.model.SCANVI.from_scvi_model(
scvi_model,
adata=adata,
unlabeled_category="Unknown",
labels_key=SCANVI_CELLTYPE_KEY,
)
scanvi_model.train(max_epochs=20, n_samples_per_label=100)
INFO Training for 20 epochs.
Epoch 20/20: 100%|██████████| 20/20 [00:32<00:00, 1.60s/it, v_num=1, train_loss_step=947, train_loss_epoch=1.03e+3]
Now we can predict the missing cell types, and get the latent space
SCANVI_LATENT_KEY = "X_scANVI"
SCANVI_PREDICTION_KEY = "C_scANVI"
adata.obsm[SCANVI_LATENT_KEY] = scanvi_model.get_latent_representation(adata)
adata.obs[SCANVI_PREDICTION_KEY] = scanvi_model.predict(adata)
Again, we may visualize the latent space as well as the inferred labels
SCANVI_MDE_KEY = "X_mde_scanvi"
adata.obsm[SCANVI_MDE_KEY] = mde(adata.obsm[SCANVI_LATENT_KEY])
INFO Using cuda:0 for `pymde.preserve_neighbors`.
# in order to make colors matchup
adata.obs.C_scANVI = pd.Categorical(
adata.obs.C_scANVI.values, categories=adata.obs.cell_ontology_class.cat.categories
)
sc.pl.embedding(
adata,
basis=SCANVI_MDE_KEY,
color=["cell_ontology_class", SCANVI_PREDICTION_KEY],
ncols=1,
frameon=False,
palette=adata.uns["cell_ontology_class_colors"],
)

Now we can observe scANVI’s performance using a confusion matrix.
df = (
adata.obs.groupby(["cell_ontology_class", SCANVI_PREDICTION_KEY])
.size()
.unstack(fill_value=0)
)
conf_mat = df / df.sum(axis=1).values[:, np.newaxis]
plt.figure(figsize=(8, 8))
_ = plt.pcolor(conf_mat)
_ = plt.xticks(np.arange(0.5, len(df.columns), 1), df.columns, rotation=90)
_ = plt.yticks(np.arange(0.5, len(df.index), 1), df.index)
plt.xlabel("Predicted")
plt.ylabel("Observed")
Text(0, 0.5, 'Observed')
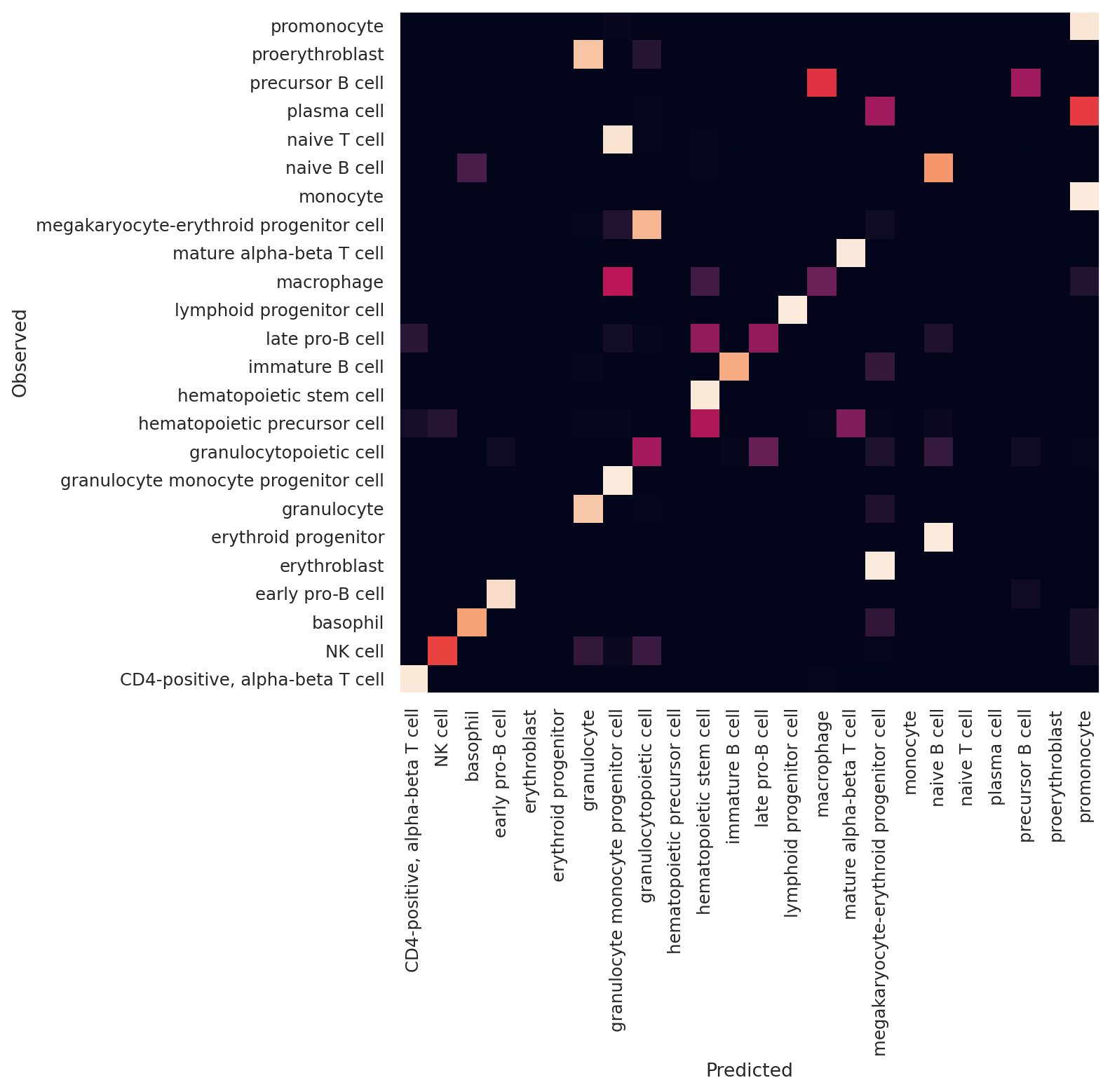
As we see in this, scANVI’s latent space separates all cell types and performs well at classifying major cell types. Since the 10X data is labelled at lower resolution, the transferred labels are not always identical with the original label. However, biologically, the transferred labels are subsets of the original labels.

